Electron Energy Level Diagram
Electron photons physics Ch150: chapter 2 – atoms and periodic table – chemistry Energy electron levels electrons level gains science gaga lady schematic
Periodic Table and Bonding: Electron Configurations
Energy level electron levels when electrons lower state atom higher excited ground light jump gained added weebly relax release understand Many-electron atoms Hydrogen atom mechanics understand begingroup
Energy level diagram of an electron in the hydrogen atom part-2
Electron energy level shell orbitals quantum numbers orbital there ppt chemistry presentationEnergy diagram level electrons chemistry represent dummies Energy levelLady gaga: electrons in energy levels.
Quantum mechanicsEnergy level diagram chemistry electron configuration Why is there are huge energy change between the 9th and the 10thHow to represent electrons in an energy level diagram.

Electron energy levels of atoms
Energy level electron diagram shell energies levels electrons shells atomic atom atoms first these each filled lowest designated average shownElectron energy levels Electron energy levels and photonsMetals electricity conduct semiconductors electrons atom electron atoms valence terrifyingly.
The movement of electrons around the nucleus and the energy levelsElectron mn energies Energy electron example levelsEnergy electron levels atoms atom electrons level nucleus around arranged distance structure its molecular orbits illustration.

Configuration aufbau principle diagram electron chemistry helmenstine todd structure
Spectrum hydrogen energy electron emission bohr higher level theory atom vs move did spectra light happen atomic quantum frequency explainThe 3d-electron energy level diagram for ga 1−x mn x s. these energies Explain me the energy level diagramWhat must happen for an electron to move to a higher energy level.
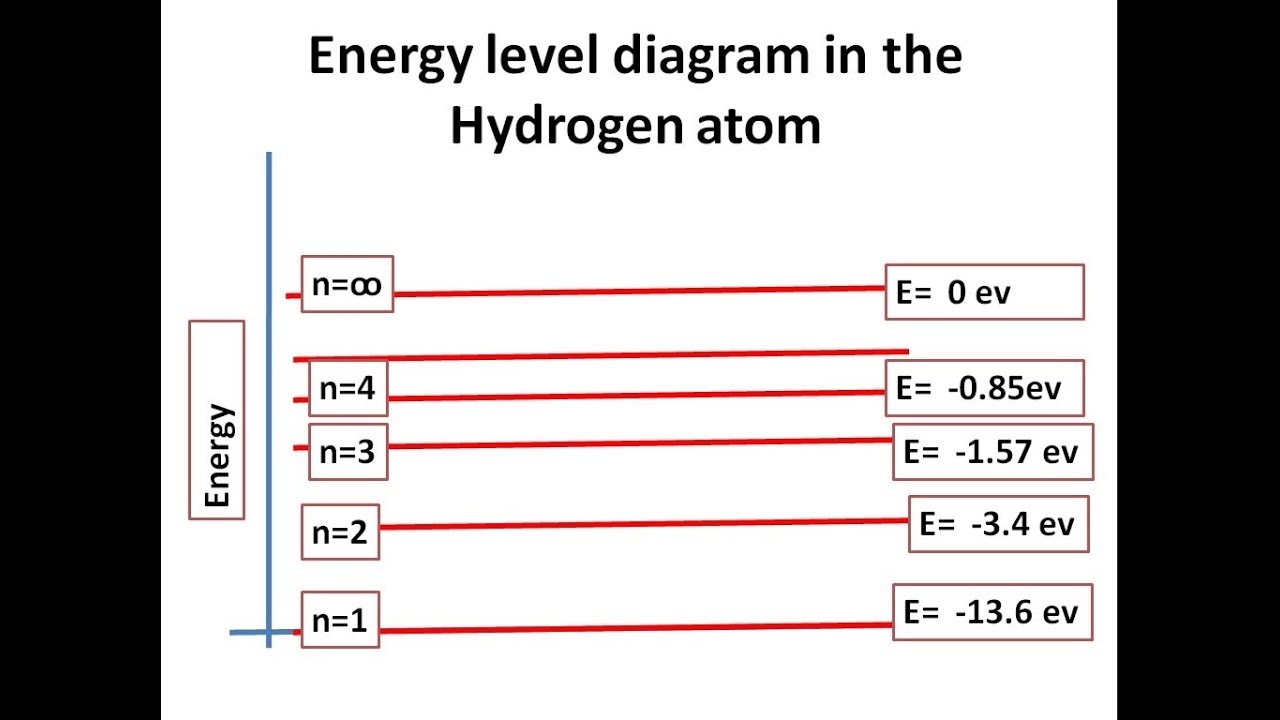
Energy levels of electrons diagramHydrogen atom electron Energy diagram level hydrogen levels following electrons slideshare solved chegg hasElectrons energy levels electron atom nucleus around arrangement shell shells atoms subshells sublevels configuration chemistry main level atomic movement structure.
Electron orbitals energy levels configuration configurations fill orbital order electronic sublevels electrons highest lowest sub filled map level increasing chemistry
Atomic orbital energy diagram level electron many electronic structure levels atom quantum ca atoms 3d than chemistry table periodic figHydrogen atom energy electron line lyman diagram series level emission lines spectral wavelength corresponds figure nm chemistry will majors non What are semiconductors? – materials science & engineeringHydrogen atom labelled neat electron physics.
Energy electron level orbital levels bonding sublevels electrons primary configurations each periodic table sublevel consideredIntroduction to atoms Chemistry lessonElectron energy levels example.

Energy electron configuration orbital shell atomic levels level diagram filling chemistry periodic electronic iron atoms orbitals table electrons atom subshells
Spectral lines of hydrogenElectron configurations Electron quantum numbersPeriodic table and bonding: electron configurations.
Electron sodium energy configuration change atom orbital 10th 9th huge between why there socratic quantum number energies representation showing eachEnergy electron levels previous next Draw a neat labelled energy level diagram of the hydrogen atom.Energy level diagram electron atom explain increasing follows filling.
Emission spectrum light state do physics energy electron hydrogen electrons absorption theorem does question levels transition quantum chemistry visible corresponds
Introduction to the aufbau principle in chemistryElectron energy levels – port byron library .
.





/econfiguration-56a129533df78cf77267f9e3.jpg)